From biomolecules to geomolecules |
Early diagenesis
|
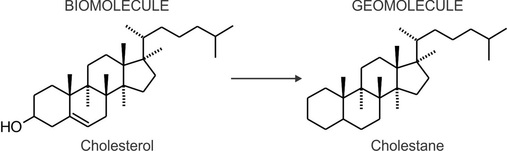
Upon death of the primary producers and heterotrophic organisms begin a transformation (diagenesis) that will lead to the destruction of their most labile compounds, and for the recalcitrant ones, the progressive disappearance of chemical functions such as hydroxyl groups, ketones, and unsaturations. The classic example of such a process is the loss the alcohol group and unsaturation of the steroid cholesterol (a biomolecule), resulting in the hydrocarbon skeleton cholestane (a geomolecule).
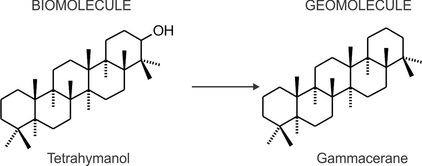
Similarly, the ciliate derived tetrahymanol loses its functions to form the hydrocarbon skeleton gammacerane, fig. 2).
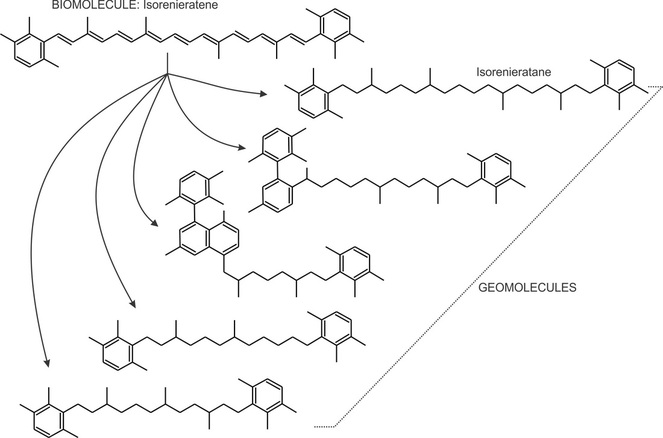
Diagenesis may also induce the full saturation of the Chlorobiaceae pigment isorenieratene to isorenieratane. It can also induce cyclization of isorenieratene, which also can lose C7 or C8 moeities to form C32 and C33 diaryl isoprenoids.
|
Further Reading |
|